Limit on lab-grown human embryos dropped by stem-cell body
By Nidhi Subbaraman,
Nature
| 05. 26. 2021
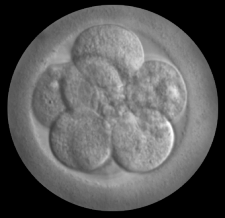
The International Society for Stem Cell Research relaxed the famous 14-day rule on culturing human embryos in its latest research guidelines.
The international body representing stem-cell scientists has torn up a decades-old limit on the length of time that scientists should grow human embryos in the lab, giving more leeway to researchers who are studying human development and disease.
Previously, the International Society for Stem Cell Research (ISSCR) recommended that scientists culture human embryos for no more than two weeks after fertilization. But on 26 May, the society said it was relaxing this famous limit, known as the ‘14-day rule’. Rather than replace or extend the limit, the ISSCR now suggests that studies proposing to grow human embryos beyond the two-week mark be considered on a case-by-case basis, and be subjected to several phases of review to determine at what point the experiments must be stopped.
The ISSCR made this change and others to its guidelines for biomedical research in response to rapid advances in the field, including the ability to create embryo-like structures from human stem cells. In addition to relaxing the ‘14-day rule’, for instance, the group advises against editing genes in human embryos until the safety of genome...
Related Articles
By Rob Stein, NPR | 04.23.2026
The Food and Drug Administration approved the first gene therapy to restore hearing for people who were born deaf.
The decision, while only immediately affecting people born with a very rare form of genetic deafness, is being hailed as...
By Marcelo Jauregui-Volpe, Wired | 04.24.2026
Two companies that launched last year with plans to create gene-edited babies have already shut down, citing money issues and internal conflict.
One of them, Manhattan Genomics of New York, closed abruptly shortly after announcing a team of scientific advisers...
By Alexandre Piquard, Le Monde [cites Katie Hasson] | 04.27.2026
"Si on en prouve la sûreté, nous croyons que l’édition préventive du génome pourrait être l’une des technologies de santé les plus importantes du siècle. » Lucas Harrington explique ainsi le but de son entreprise Preventive : créer des bébés génétiquement modifiés...
By Abby Vesoulis, Mother Jones | 04.18.2026
Two years ago, we devoted an entire issue to the rise of the American oligarchy. Since then, our oligarchic system has become more entrenched and pervasive, revolving around a small crew of tech titans whose quest for wealth and...