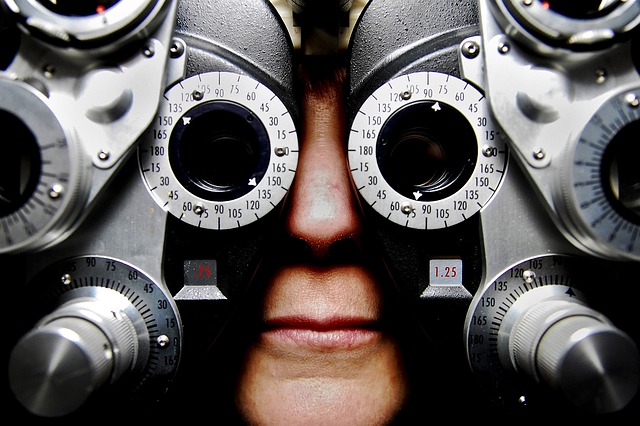
Groundbreaking CRISPR treatment for blindness only works for subset of patients
By Jocelyn Kaiser,
Science
| 11. 17. 2022
After some early but cautious optimism, a company is shelving its pioneering gene-editing treatment for a rare inherited blindness disorder. Editas Medicine announced today the trial trying to use the gene editor CRISPR to treat Leber congenital amaurosis 10 (LCA10) led to “clinically meaningful” vision improvements in only three of 14 patients.
In the study, patients received a subretinal injection of a modified virus carrying genetic material encoding components of CRISPR, a DNA-cleaving enzyme, and two RNA strands to guide the protein to its target sequences. For this trial, CRISPR was designed to snip out a problematic part of a gene called CEP290, which encodes a protein needed by the photoreceptor cells that eyes use to sense light. It was the first trial in the world to directly inject the gene-editing system into the body, rather than applying CRISPR to people’s cells in a lab dish and then reinfusing them.
Two of the patients whose vision improved—a woman treated early in the trial and a 14-year-old boy treated more recently, both of whom received midrange doses—had two defective copies...
Related Articles
By Dr. Coco Newton, Progress Educational Trust | 03.30.2026
Have you ever wondered what it means to have dozens of half-siblings across the world – or to never know where half of your genetic identity comes from? A recent episode of Zembla explores the human consequences of the global...
By Rob Stein, NPR | 04.23.2026
The Food and Drug Administration approved the first gene therapy to restore hearing for people who were born deaf.
The decision, while only immediately affecting people born with a very rare form of genetic deafness, is being hailed as...
By Emily Mullin, Wired | 04.23.2026
A STARTUP OUT of Utah, Paterna Biosciences, says it has successfully grown functional human sperm in a lab and used the sperm to make visibly healthy-looking embryos. The technique could eventually help men with certain types of infertility have biological children...
By Julianna LeMieux, Genetic Engineering & Biotechnology News | 04.14.2026
Twenty years ago, Sven Bocklandt, PhD, sought to create a hypoallergenic cat. He had the genetic engineering chops to do it, but the embryology was beyond his capabilities. At a small animal genetic engineering conference, known as TARC (Transgenic Animal...