Choosing Life With Down Syndrome
By Ruth Graham,
Slate
| 05. 31. 2018
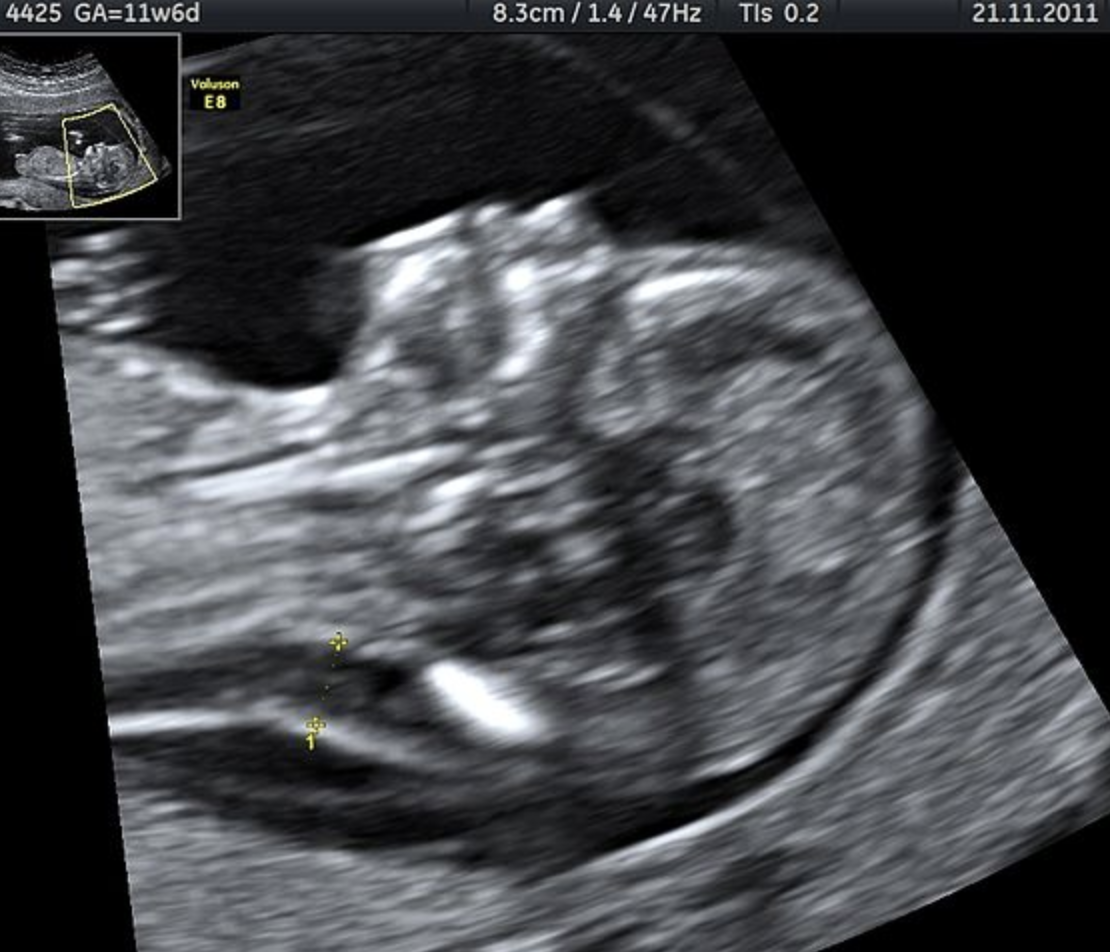
Celeste Blau’s first pregnancy was smooth from the beginning. She stayed active and kept working at the gelato shop she owns with her family outside of Cleveland. She wasn’t ignoring the pregnancy, exactly—it was planned—but she didn’t think of herself as a very maternal person, and pregnancy seemed like something to endure rather than to enjoy. At her first ultrasound, she was mostly excited to find out whether she was having a boy or a girl. She was 31, relatively young, and it didn’t occur to her that anything could be wrong.
The ultrasound appointment at the Cleveland Clinic took three hours. She only realized that was unusual a few years later, when she was pregnant again and the same process took 30 minutes. Lying on the table, her belly smeared with gel, she started to feel a niggling fear when the technicians weren’t laughing at her and her husband’s jokes. (“And we’re funny!” she said.) Afterward, the doctor asked to speak to them in his office. He was concerned because the baby’s brain ventricles looked enlarged, Blau recalls, and...
Related Articles
By Abby Vesoulis, Mother Jones | 04.18.2026
Two years ago, we devoted an entire issue to the rise of the American oligarchy. Since then, our oligarchic system has become more entrenched and pervasive, revolving around a small crew of tech titans whose quest for wealth and...
By Miguel Muñoz, Cadena SER [cites Marcy Darnovsky] | 08.04.2026
"Para ellos, una familia numerosa no solo es una preferencia personal, sino que es una obligación. Creen que tener tantos hijos como sea posible es necesario para evitar un futuro apocalíptico", aseguraba Xavier Orri, periodista y cofundador de Página Internacional...
By Ryan Cross, Endpoints News | 03.24.2026
Cathy Tie has an audacity more typical of a tech startup founder than a biotech executive. She dropped out of college to start a genetic screening company and later founded a telemedicine startup. The 29-year-old has been on two Forbes...
By Alex Polyakov, The Conversation | 02.09.2026
Prospective parents are being marketed genetic tests that claim to predict which IVF embryo will grow into the tallest, smartest or healthiest child.
But these tests cannot deliver what they promise. The benefits are likely minimal, while the risks to...