Theranos and COVID-19 Testing Are Mirror-Image Cautionary Tales
By Benjamin Mazer,
The Atlantic
| 09. 15. 2021
Photo by Steve Nomax on Unsplash
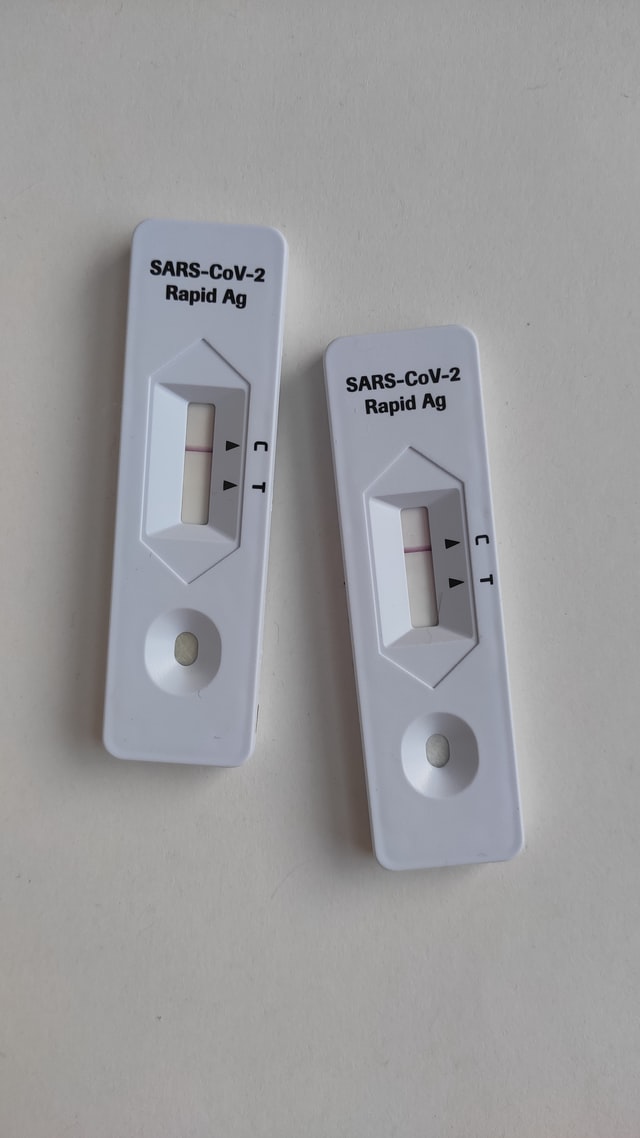
Last week, prosecutors and defense attorneys made opening statements in the criminal trial of Elizabeth Holmes, the former CEO of Theranos, who is accused of defrauding investors and patients with false promises of cheap, rapid blood tests. The next day, the Biden administration announced a plan to purchase 280 million cheap, rapid COVID-19 tests—an action for which some lawmakers have been advocating for more than a year. The serendipity of these two events exposes an unresolved tension in health care: How do we balance the risks of disruptive innovation with the mortal costs of administrative inertia?
Theranos and COVID-19 testing are both cautionary tales of failed medical oversight, but the morals flip from one case to the other. Each addresses, in its way, how much control the FDA should exert over laboratory tests before they come to market. COVID-19 testing epitomizes the agency’s risk-averse instincts. In the first months of the coronavirus pandemic, the FDA notoriously delayed private labs from developing their own COVID-19 tests. This allowed the coronavirus to spread mostly...
Related Articles
By Staff, ABC News | 06.01.2026
The Victorian government is introducing legislation it says will make IVF clinics safer and more accountable following high-profile bungles by private providers.
As part of the changes, the state's health minister will have the power to personally intervene to cancel...
By Sofia Resnick, Stateline | 05.20.2026
An anti-abortion group last month sued seven Utah fertility clinics, claiming their disposal of embryos as part of the in vitro fertilization process violates the state’s wrongful death law.
The ministry Voice for the Voiceless believes it has a strong...
By Laura Hughes, Financial Times | 05.20.2026
Sophie and her husband are set to spend more than £100,000 in travel and medical bills as they fly between England and the US in their bid to have another child.
The couple are undergoing IVF treatment in New York...
By Sofia Bettiza, BBC News | 05.07.2026
Karina is six months pregnant, but the foetus inside her womb is not her own.
The 22-year-old from eastern Ukraine is a surrogate, pregnant with an embryo from a Chinese couple's egg and sperm.
At the age of 17 Karina's...