New way for states to cover pricey gene therapies will start with sickle cell disease
By Nada Hassanein,
New Jersey Monitor
| 03. 14. 2024
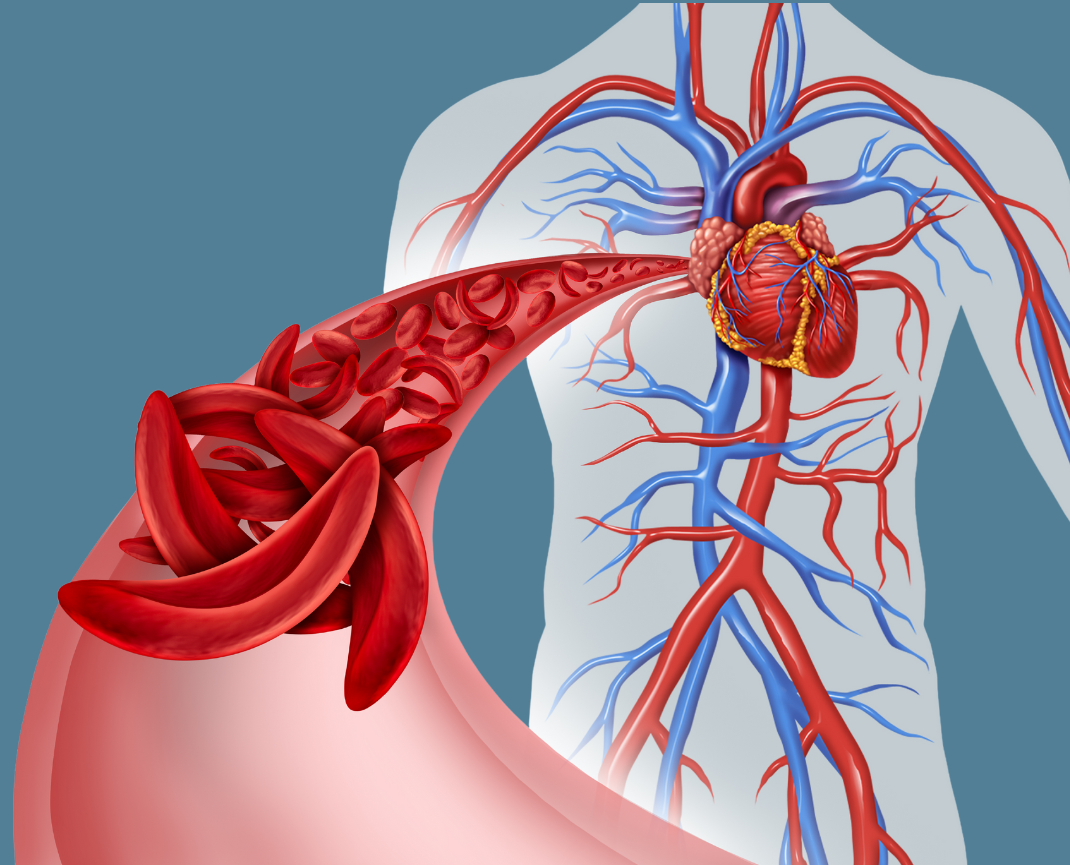
The U.S. Food and Drug Administration late last year approved two breakthrough gene therapies for sickle cell disease patients. Now a new federal program seeks to make these life-changing treatments available to patients with low incomes — and it could be a model to help states pay for other expensive therapies.
The new sickle cell treatments have brought hope to those with the debilitating blood disorder, which is hereditary and disproportionately affects Black people. But the therapies come with a price tag of as much as $3 million for a course of treatment, which can take up to a year. Despite those high upfront costs, cell and gene therapies have the potential to reduce health care spending over time by addressing the underlying cause of the disease.
Under the so-called Cell and Gene Therapy Access Model, the federal government will negotiate discounts with sickle cell drug manufacturers Vertex Pharmaceuticals, CRISPR Therapeutics and Bluebird Bio on behalf of state Medicaid agencies, which provide health care coverage to low-income patients. To participate, state Medicaid agencies must agree to prices based on those negotiations...
Related Articles
By Rob Stein, NPR | 04.23.2026
The Food and Drug Administration approved the first gene therapy to restore hearing for people who were born deaf.
The decision, while only immediately affecting people born with a very rare form of genetic deafness, is being hailed as...
By Marcelo Jauregui-Volpe, Wired | 04.24.2026
Two companies that launched last year with plans to create gene-edited babies have already shut down, citing money issues and internal conflict.
One of them, Manhattan Genomics of New York, closed abruptly shortly after announcing a team of scientific advisers...
By Alexandre Piquard, Le Monde [cites Katie Hasson] | 04.27.2026
"Si on en prouve la sûreté, nous croyons que l’édition préventive du génome pourrait être l’une des technologies de santé les plus importantes du siècle. » Lucas Harrington explique ainsi le but de son entreprise Preventive : créer des bébés génétiquement modifiés...
By Abby Vesoulis, Mother Jones | 04.18.2026
Two years ago, we devoted an entire issue to the rise of the American oligarchy. Since then, our oligarchic system has become more entrenched and pervasive, revolving around a small crew of tech titans whose quest for wealth and...