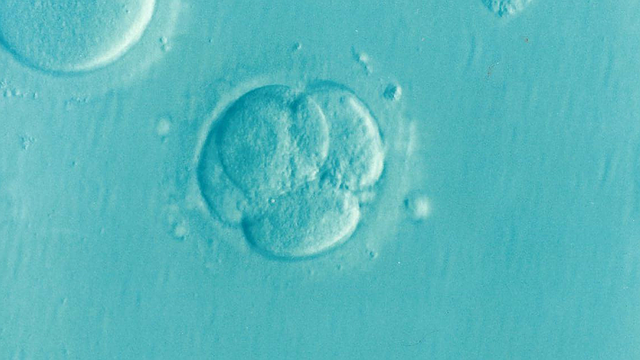
A Gene Editing Breakthrough
By Tom Ashbrook,
NPR On Point [features Marcy Darnovsky]
| 08. 08. 2017
After the blockbuster announcement a U.S. team successfully edited human embryos come the tough medical and ethical questions. We’ll talk it through.
A first in the U.S.: researchers have now edited the genetic code of viable human embryos, cutting and splicing to avoid defect and disease. The DNA of human embryos. Genetically edited. The edited embryos weren’t implanted in a woman. Weren’t born. But they could have been. New life spared disease, maybe death. How far does this gene editing go? How fast? For whom? And for, or against, what human traits? This hour On Point: editing the code of human life. -- Tom Ashbrook
Guests
Amy Dockser Marcus, health and science reporter for the Wall Street Journal. (@AmyDMarcus)
Dr. Paula Amato, co-author of the study showing successful editing of genes in human embryos. Associate professor of obstetrics and gynecology at Oregon Health & Science University.
Arthur Caplan, professor of bioethics and head of the division of medical ethics at the New York University School of Medicine. (@ArthurCaplan)
Marcy Darnovsky, executive director of...
Related Articles
By Rob Stein, NPR | 04.23.2026
The Food and Drug Administration approved the first gene therapy to restore hearing for people who were born deaf.
The decision, while only immediately affecting people born with a very rare form of genetic deafness, is being hailed as...
By Marcelo Jauregui-Volpe, Wired | 04.24.2026
Two companies that launched last year with plans to create gene-edited babies have already shut down, citing money issues and internal conflict.
One of them, Manhattan Genomics of New York, closed abruptly shortly after announcing a team of scientific advisers...
By Alexandre Piquard, Le Monde [cites Katie Hasson] | 04.27.2026
"Si on en prouve la sûreté, nous croyons que l’édition préventive du génome pourrait être l’une des technologies de santé les plus importantes du siècle. » Lucas Harrington explique ainsi le but de son entreprise Preventive : créer des bébés génétiquement modifiés...
By Abby Vesoulis, Mother Jones | 04.18.2026
Two years ago, we devoted an entire issue to the rise of the American oligarchy. Since then, our oligarchic system has become more entrenched and pervasive, revolving around a small crew of tech titans whose quest for wealth and...